In a recent study published in the BMC Medicine journal, researchers in Sweden explored the association between above-optimal maternal gestational weight gain (GWG) and the risk of neurodevelopmental disorders such as intellectual disability, autism spectrum disorder (ASD), and attention-deficit/hyperactivity disorder (ADHD) in offspring.
 Rates of maternal weight gain over the course of pregnancy and offspring risk of neurodevelopmental disorders. Image Credit: Hazal Ak / Shutterstock
Rates of maternal weight gain over the course of pregnancy and offspring risk of neurodevelopmental disorders. Image Credit: Hazal Ak / Shutterstock
Background
Neurodevelopmental disorders are highly prevalent, and the social support requirements throughout the affected individual’s life significantly burden their families. The three most prevalent neurodevelopmental disorders —ASD, ADHD, and intellectual disability — often present together in children.
Furthermore, while de novo and inherited mutations have both been associated with these neurodevelopmental disorders, other environmental, biological, and social factors are also thought to contribute to their etiology.
Although previous studies have explored the association between maternal GWG beyond the optimal range and the elevated risk of neurodevelopmental disorders, it has been difficult to separate the effects of above-optimal GWG and gestational duration on the adverse outcomes related to neurodevelopmental disorders. This is because these studies did not consider the length of the pregnancy as a determinant factor.
Since the growth of the functional and structural parts of the fetal brain is a sequential process and the vulnerability to external factors such as nutrition and environmental stressors varies across trimesters, it is vital to assess the link between trimester-specific GWG and the risk of neurodevelopmental disorders.
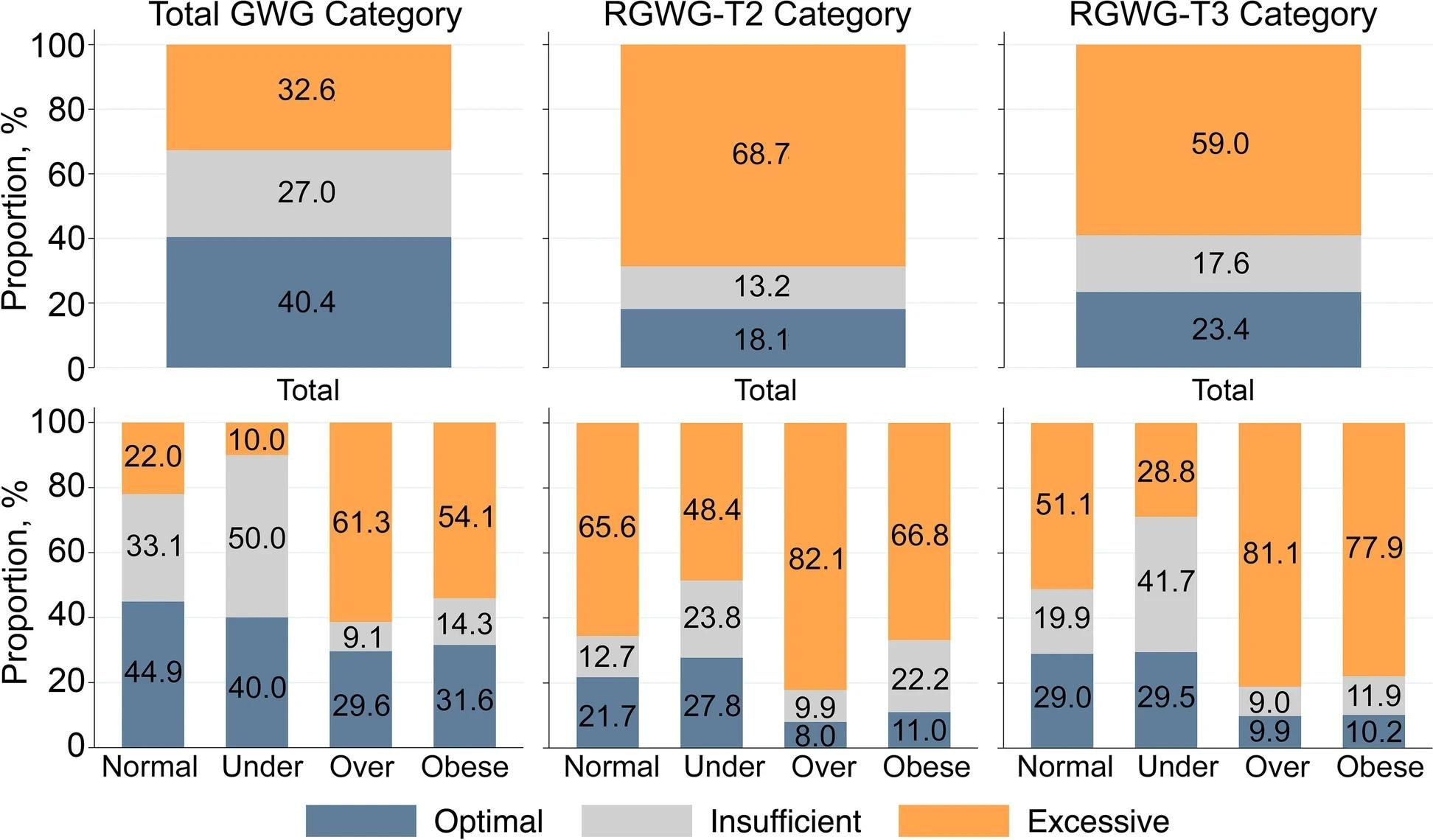 Distributions of total GWG (kg), RGWG-T2, and RGWG-T3 categories according to the IOM guidelines
Distributions of total GWG (kg), RGWG-T2, and RGWG-T3 categories according to the IOM guidelines
About the study
In the present study, the researchers conducted a cohort study among the Swedish population to evaluate the risk of neurodevelopmental disorders according to the z-scores for GWG and the rate of weight gain during the last two trimesters of the pregnancy.
They used data from Stockholm’s record system for antenatal care and information on outcomes, exposures, and covariates from regional and national health and administrative registries. Children born between January 2007 and December 2010 for whom the data on maternal GWG measurements were available were included in the study.
All neurodevelopmental disorder diagnoses were considered for the primary analysis, including one or multiple diagnoses of ADHD, intellectual disability, and ASD. For the secondary analysis, the researchers only included combinations of mutually exclusive outcomes such as only ADHD or ASD, ADHD with ASD but no intellectual disability.
The examined exposures comprised total GWG and the rate of GWG for the second and third trimesters of the pregnancy. The analysis was also conducted using the “insufficient,” “optimal,” and “excessive” categories for the rate of GWG for each body mass index group. Potential confounding covariates included birth year, sex of the child, household income, paternal and maternal region of birth, maternal age, mother’s education level, interpregnancy interval, psychiatric history of the mother, and smoking behavior.
Results
The results indicated that higher than optimal total GWG was associated with a 19% increase in the risk of any neurodevelopmental disorder, while lower than optimal GWG increased the risk of neurodevelopmental disorders by 12%.
Furthermore, the rate of GWG was also associated with the risk of neurodevelopmental disorders, with a slower GWG rate during the second trimester increasing the risk of neurodevelopmental disorders by 9%, but a higher rate of GWG in the second trimester not being associated with neurodevelopmental disorder risk.
In contrast, while a slower rate of GWG was not linked to neurodevelopmental disorder risk in the third trimester, a higher rate of GWG was associated with a 28% increase in the risk of neurodevelopmental disorder diagnoses.
In the secondary analysis using categorized rates of GWG in the last two trimesters, the results reported that a low GWG rate in the second trimester combined with an excessive rate of GWG in the third trimester significantly increased the risk of intellectual disability and ADHD in the offspring.
The authors also discussed plausible mechanisms for linking excessive GWG and fetal neurodevelopment, such as the downstream impact of maternal and fetal adipose tissue accumulation.
Increased adiposity in the mother and fetus is thought to be connected to dysregulations of the pro-inflammatory cytokine, insulin, leptin, and glucose signaling, increased oxidative stress, and dysregulated signaling related to dopamine and serotonin. In contrast, insufficient GWG could result in a nutrition deficit environment, which has detrimental effects on the fetus’s brain development.
Conclusions
Overall, the results provided evidence that maternal GWG below and above the optimal range were associated with an increased risk of neurodevelopmental disorders in the offspring. The results also indicated that the rate of GWG was also an important factor in determining neurodevelopmental disorder risk.
Insufficient GWG rate during the second trimester coupled with a rapid rate of GWG in the third trimester was linked to the highest risk of neurodevelopmental disorders, especially ADHD and intellectual disability.
These findings suggested that the rate of maternal GWG could potentially be used as a marker to estimate the risk of neurodevelopmental disorders in the fetus.