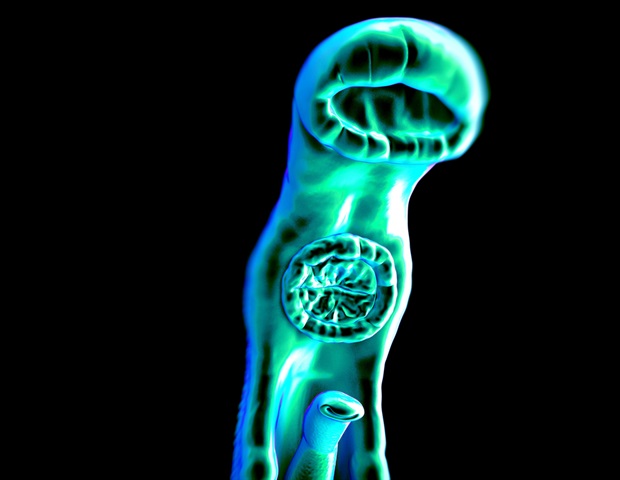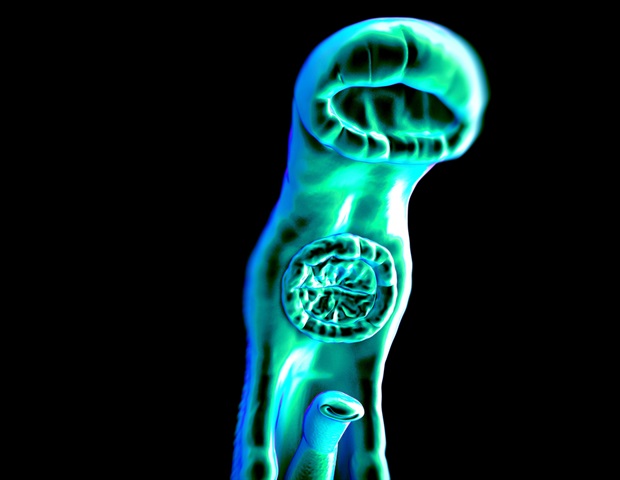
The worms that cause schistosomiasis (Schistosoma mansoni) are unusual in several ways, especially the fact that male and female adults must stay paired together throughout their lives for reproduction to be successful. Females may produce as many as 3,000 eggs per day. Approximately half reach the host’s gut or bladder. The rest are swept away via the blood to the liver and spleen, where they cause severe inflammation and liver cirrhosis, the main cause of mortality.
Researchers at Butantan Institute in São Paulo, Brazil, discovered a way to separate males from females, preventing reproduction and egg release. In an article published in PLOS Pathogens, they describe how they achieved this separation by silencing long non-coding RNAs, which are therefore a promising target for treating the disease.
Long non-coding RNAs (lncRNAs) are generally defined as transcripts with size larger than 200 nucleotides that are not translated into protein.
“For many years, lncRNAs were overlooked by researchers despite the fact that they account for 97% of all the RNA in human cells, because they had no known functions. In the last two decades, cancer research above all has shown that when lncRNAs are dysregulated, they can cause disease. Our study shows for the first time and in a functional manner that lncRNAs are essential to maintain homeostasis in the parasite that causes schistosomiasis and are therefore potential therapeutic targets,” said Murilo Sena Amaral, co-corresponding author of the article and a researcher at Butantan Institute’s Cell Cycle Laboratory.
The discovery was part of a Thematic Project, supported by FAPESP, to investigate the role of lncRNAs generally, using human cancer and Schistosoma worms as models. The principal investigator is Sergio Verjovski-Almeida, a professor at the University of São Paulo (USP) and a researcher at Butantan Institute. FAPESP also supported the study via four other projects (18/24015-0, 19/09404-3, 18/19591-2 and 16/10046-6).
Here it may be useful to recall that in humans, plants and animals (including parasites), all genetic information is contained in DNA, which serves as a sort of mold for the transcription of RNA in the cell nucleus. Verjovski-Almeida stressed that this sequence of events is known as the “central dogma of molecular biology”: genetic information flows only in one direction, from DNA to RNA to protein, and proteins perform all kinds of function in cells. Most of the RNA transcribed does not translate into protein but plays vital roles in cellular processes, as research has shown in recent decades.
The researchers analyzed data from public repositories to identify the lncRNAs from S. mansoni that were most or least expressed when males and females were paired or separate and then used the results to select three lncRNAs as candidate therapeutic targets.
“S. mansoni is well-adapted to living in the host’s mesenteric veins [which perfuse the intestines] and can remain there for decades if there is no treatment. Pairing – the female living inside the male – is essential to their survival. Without it, they die, as we proved in our laboratory experiments,” Amaral said.
Separation and death
The researchers began with in vitro assays, placing pairs of male and female worms in culture dishes containing a medium with blood, and adding a molecule capable of targeting the lncRNA of interest so as to reduce it in the parasite.
“For proof of concept we used a double-stranded RNA molecule,” Amaral explained. “When added to the culture medium, it binds to the lncRNA in the parasite and leads to its degradation. After a time, we found that the parasites that received the treatment separated, became less viable, stopped releasing eggs and died.”
Next, the researchers conducted experiments in mice infected by S. mansoni. They injected the same double-stranded RNA into the animal’s bloodstream, and over time the target lncRNA decreased in the parasites, leading to their death and diminishing the viability of their eggs.
Neglected disease
Schistosomiasis is the main disease caused by helminths (parasitic worms), affecting some 200 million people worldwide. Despite this significant prevalence, for 40 years praziquantel has been the only drug available to treat the disease.
According to Verjovski-Almeida, praziquantel has major limitations. “It’s been on the market for a long time without any alternatives, and there are reports of resistant worms. Hence the need to find novel therapeutic targets against the disease. Our study proved that the parasites can be eliminated from the host’s bloodstream by attacking the pairing phenomenon. Our next step is to develop a drug that can do what the double-stranded RNA did in our study: silence expression of the lcnRNA in the parasite,” he said.
Source:
São Paulo Research Foundation (FAPESP)
Journal reference:
DOI: 10.1371/journal.ppat.1011369